|
Modern Periodic Law - The properties of the elements of the modern periodic law are periodic functions of their atomic numbers. Although he predicted the properties of those elements through his periodic classification of elements. He left some space for corresponding elements in his periodic table which were not even discovered till then. Mendeleev’s Periodic Table - The arrangement of all 63 elements in rows or columns in order of their atomic weight was made by Mendeleev. It must be noted, atoms lack a well-defined outer boundary. Newlands Law of Octaves - The elements were arranged in increasing order of their atomic weights and found that every 8 th element shows similarity with the 1 st element. The atomic radius of Aluminium atom is 121pm (covalent radius). In every triad, the atomic weight of the middle element was equal to the average of the atomic weights of the first and third elements. Highest ionization energy Lowest ionization energy Answer Bank TP IN Sb As IĪrrange the species in order of decreasing first ionization energy.Since many elements were being discovered in the 19 th century and the study of these elements individually was proving difficult, classification of elements was made necessary.Ĭlassification by Johann Dobereiner - German chemist Johann Dobereiner classified certain elements on the basis of their similar properties in the groups of continuing - three elements each. Highest ionization energy Lowest ionization energy Answer Bank Rb Test Xe || SnĪrrange these elements according to first ionization energy. The atomic radius of rare earth element is 0.174-0.204 nm 12, which is larger than the atomic radius of aluminum (0.143 nm). | Rank these elements according to first ionization energy. O cannot be determined based on periodic trends Oc OP Predict which element has the larger first ionization energy based on periodic trends. O cannot be determined based on periodic trends OK 0 Mg Predict which element has the larger first ionization energy based on periodic trends. Predict which element has the larger first ionization energy based on periodic trends. Highest ionization energy Lowest ionization energy Answer Bank KC A10 Os²- O Ca²+Īrrange the elements according to their firs ionization energies. 2 Vincent D The spectral radius of a finite graph is defined to be the spectral radius of its adjacency matrix Jurga´s (Czornik, 2005) Projection-valued measures 48 3 As corollaries of our first result, two previous theorems due to Fiedler and Nikiforov and Lu et al As corollaries of our first result, two previous theorems due to Fiedler and Nikiforov and. OF OF Determine which is the larger species. This site offers comprehensive information for each element including: who, when & where up to 40 properties (chemical & physical) over 3,600 nuclides (isotopes) over 4,400 nuclide decay modes the element names in 10 different languages and more. O Na O Na+ Determine which is the larger species. Largest Smallest Answer Bank oxygen silicon calcium barium Arrange these elements according to atomic radius. Highest ionization energy Lowest ionization energy Answer Bank Se s o Te Teĭetermine which is the larger species. Largest radius Smallest radius Answer Bank Ar Si Mg Al Na Arrange the elements according to atomic radius, from largest to smallest. 
Highest ionization energy Lowest ionization energy Answer Bank Ne Be LiĪrrange the elements according to first ionization energy. Largest radius Smallest radius Answer Bank p3- K+ s2- Cr Ca2+Īrrange these elements according to first ionization energy. Largest radius Smallest radius Answer Bank Br+ Brº BrĪrrange these ions according to ionic radius. Largest radius Smallest radius Answer Bank Li | Lit | LiĪrrange the atom and ions from largest to smallest radius. Largest radius Smallest radius Answer Bank Be Mg Mg Rb Ca СаĪrrange the atom and ions according to radius. This diameter between the atoms is divided by two to give the radius. Download : Download high-res image (175KB) Download : Download full-size image Fig. 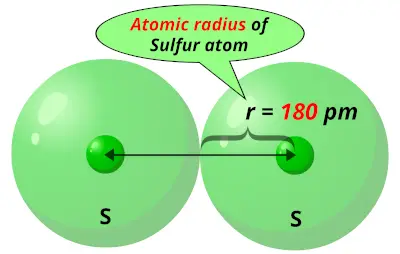
Largest Smallest Answer Bank oxygen silicon calcium bariumĪrrange these elements according to atomic radius. The atomic radius is measured based on the distance between the nuclei of two atoms that are barely touching each other, which means the electron shells of the two atoms are just touching each other. Another explanation is the large atomic radius of Al (the atomic radius of alloying elements is shown in Table 4), and the addition of Al atoms will intensify the lattice distortion, which has obvious strengthening effect of solution in alloy system. Largest radius Smallest radius Answer Bank Ar Si Mg Al NaĪrrange the elements according to atomic radius, from largest to smallest. Transcribed image text: Arrange the elements according to atomic radius.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
